News Archive
Novel insights into the replication mechanisms of the influenza virus
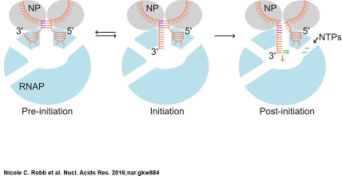
October 2016. X-ray structures of the influenza virus RNA polymerase have provided intriguing static snapshots of how the viral RNA genome is bound and copied. Research teams from Oxford and Frankfurt have extended these studies by using a solution-based single-molecule FRET assay to probe dynamic motions of the RNA that occur during polymerase binding and initial replication. These single molecule studies show that even in the absence of nucleotides the polymerase-bound RNA template exists in two conformations, corresponding to pre-initiation and initiation states. Nucleotide addition stabilizes the RNA in the active site and results in unwinding of the duplexed region of the promoter. Robb and colleagues propose a model of the dynamic processes that occur during early influenza RNA synthesis and provide novel insights into the replication mechanisms of this important pathogen.
Contact:
Robert Tampé
Institute of Biochemistry, Biocenter, Goethe University Frankfurt
Max von Laue Str. 9, 60438 Frankfurt am Main, Germany
tampe@em.uni-frankfurt.de, www.biochem.uni-frankfurt.de
Reference:
Robb NC, Te Velthuis AJ, Wieneke R, Tampé R, Cordes T, Fodor E, Kapanidis AN (2016) Single-molecule FRET reveals the pre-initiation and initiation conformations of influenza virus promoter RNA. Nucleic Acids Res, http://dx.doi.org/10.1093/nar/gkw884