News
Cause of severe hereditary disease revealed
January 2018. The origin of many diseases lies in genetic abnormalities resulting in the functional defect of the proteins they are encoding. A well-known example is p53, the tumour suppressor protein. Inactivation of p53 by genomic mutations, amongst others, is one of the first essential steps in the development of tumours. Defects of the related protein p63, however, lead to a group of human disorders characterized by defects in embryonic development. Scientists of the Goethe University Frankfurt in collaboration with a research group of the University Neapel Federico II have now shed light on the so far unknown mechanism of one of these syndromes.
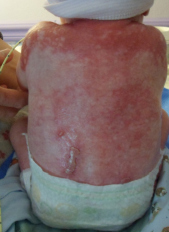 The transcription factor p63 is the master regulator of epidermal development and plays an essential role in proliferation of basal stem cells. Mutations in a certain part of p63 are causative for the life-threatening disorder ankyloblepharonectodermal defects-cleft lip/palate (AEC) syndrome, which is characterized by a cleft palate and long-lasting skin erosions similar to severe burns. While some symptoms can be treated by surgery or other medical therapy, an approach to treat the cause of the AEC syndrome has not been feasible so far due to lack of knowledge about the mutated p63’s mode of action.
The transcription factor p63 is the master regulator of epidermal development and plays an essential role in proliferation of basal stem cells. Mutations in a certain part of p63 are causative for the life-threatening disorder ankyloblepharonectodermal defects-cleft lip/palate (AEC) syndrome, which is characterized by a cleft palate and long-lasting skin erosions similar to severe burns. While some symptoms can be treated by surgery or other medical therapy, an approach to treat the cause of the AEC syndrome has not been feasible so far due to lack of knowledge about the mutated p63’s mode of action.
AEC syndrome mutations cluster in two domains of the protein and are mutually exclusive to the ones of the other syndromes associated with p63. Since these domains are known for mediating protein-protein interactions, it was assumed that loss of those interactions is responsible for the disorder. Instead, the groups of Volker Dötsch und Caterina Missero showed that mutations within p63 expose hydrophobic amino acids sequences of the protein. These sequences attach to each other forming large unstructured complexes, which leads to the loss of p63’s function as a stem cell factor. Similar types of the so-called protein aggregates are found in several other human diseases, e.g. Parkinson’s, Alzheimer’s or ALS.
A wide variety of biochemical, biophysical and cell biological methods as well as a mouse model of the disorder were required to decipher this novel mechanism in detail, a success only made possible through the close and interdisciplinary collaboration between both research groups. In their article, published in the scientific journal PNAS, the scientists also reveal that once the formation of aggregates is blocked, p63 regains its activity. This study therefore provides the foundation for the potential development of novel therapeutic approaches to combat the p63 aggregates.
Photo: Patient with AEC syndrome (photo courtesy of Virginia Sybert)
Contact:
Volker Dötsch, Buchmann Institute for Molecular Life Sciences and Institute of Biophysical Chemistry, Riedberg Campus, Goethe University Frankfurt, Germany, vdoetsch@em.uni-frankfurt.de
Publication:
Claudia Russo*, Christian Osterburg*, Anna Sirico*, Dario Antonini, Raffaele Ambrosio, Julia Maren Würz, Jörg Rinnenthal, Marco Ferniani, Sebastian Kehrloesser, Birgit Schäfer, Peter Güntert, Satrajit Sinha, Volker Dötsch* und Caterina Missero*. Protein aggregation of the p63 transcription factor underlies severe skin fragility in AEC syndrome. Proceedings of the National Academy of Sciences USA, early edition, published online 16 January 2018. http://dx.doi.org/10.1073/pnas.1713773115.

